 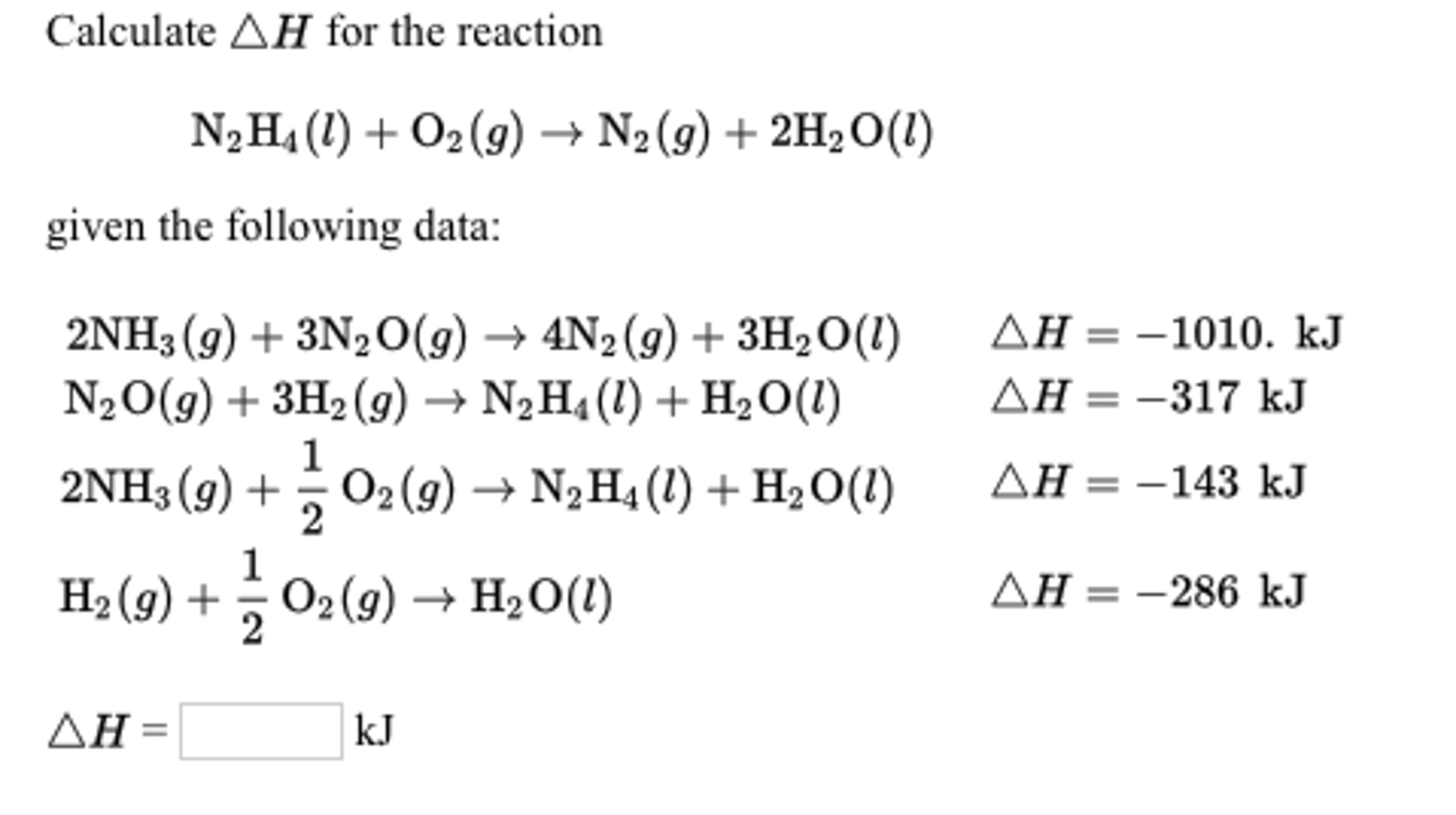
The results corrected for 1 atm pressure of H2 are 18.0 kJ/mol and -10.9 J/(mol K) Rathke, Klingler, et al., 1992 MSĪverage of 4 out of 11 values Individual data points Solvent: n-Heptane Temperature range: 307-428 K. Solvent: n-Heptane Temperature range: 353-428 K MS A rather different value has, however, been reported for the activation enthalpy of the forward reaction, 104.6 kJ/mol Ungváry, 1972 MS Solvent: n-Heptane The reaction enthalpy relies on the experimental values for the forward and reverse activation enthalpies, 72.4 and 46.0 kJ/mol, respectively Alemdaroglu, Penninger, et al., 1976. The results corrected for 1 atm pressure of H2 are 16.7 kJ/mol and -17.6 J/(mol K) Rathke, Klingler, et al., 1992 MS Solvent: Supercritical carbon dioxide Temperature range: 333-453 K. Reaction search pages in place of the enumerated reactionīy formula: C 8Co 2O 8 (solution) + H 2 (solution) = 2 C 4HCoO 4 (solution) Quantity Go To: Top, Gas phase thermochemistry data, Phase change data, Henry's Law data, Mass spectrum (electron ionization), Constants of diatomic molecules, References, Notes 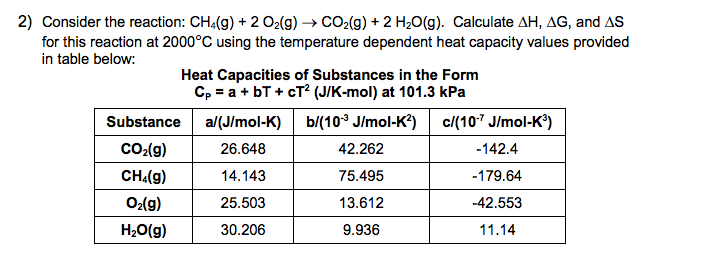
SRD 156 – Clathrate Hydrate Physical Property Database SRSD 3 – Web Thermo Tables (WTT), professional edition SRSD 2 – Web Thermo Tables (WTT), "lite" edition SRD 103b – Thermo Data Engine (TDE) for pure compounds, SRD 103a – Thermo Data Engine (TDE) for pure compounds. (TRC) data available from this site, much more physicalĪnd chemical property data is available from the In addition to the Thermodynamics Research Center mol/l by extrapolation of rectilinear diameter to Tc TRCĬoefficents calculated by NIST from author's data. Uncertainty assigned by TRC = 0.0119 bar derived from vapor pressure extrapolated to Tc TRC Uncertainty assigned by TRC = 0.2 K derived from P-V-T measurements TRC Uncertainty assigned by TRC = 0.0004 bar TRC Uncertainty assigned by TRC = 0.05 K temperature measured with He gas thermometer TRC Uncertainty assigned by TRC = 0.06 K see property X for dP/dT for c-l equil. TRC - Thermodynamics Research Center, NIST Boulder Laboratories, Chris Muzny director Go To: Top, Gas phase thermochemistry data, Reaction thermochemistry data, Henry's Law data, Mass spectrum (electron ionization), Constants of diatomic molecules, References, Notes Requires a JavaScript / HTML 5 canvas capable browser.ĭata last reviewed in March, 1977 New parameter fit October 2001 Gas Phase Heat Capacity (Shomate Equation) Secretary of Commerce on behalf of the U.S.A. Go To: Top, Phase change data, Reaction thermochemistry data, Henry's Law data, Mass spectrum (electron ionization), Constants of diatomic molecules, References, Notesīy the U.S. Your institution may already be a subscriber.įollow the links above to find out more about the data With the development of data collections included in The purpose of the fee is to recover costs associated NIST subscription sites provide data under theĭata Program, but require an annual fee to access. NIST / TRC Web Thermo Tables, professional edition (thermophysical and thermochemical data).NIST / TRC Web Thermo Tables, "lite" edition (thermophysical and thermochemical data).Computational Chemistry Comparison and Benchmark Database.Electron-Impact Ionization Cross Sections (on physics web site).Reaction thermochemistry data (reactions 1 to 50).Use this link for bookmarking this species This structure is also available as a 2d Mol file IUPAC Standard InChIKey: UFHFLCQGNIYNRP-UHFFFAOYSA-N Copy.Why? Because if you HAD brought the (1/2)O2 over from the right to the left, it would have appeared there as a -(1/2) O2 - the only way you could "MOVE IT" is to subtract it from both sides of the equation, just as you move anything around in an equation! Note that the O2 result on the left is the NET of the (3/2)O2 on the left, and the (1/2) O2 on the right, as addends. delta.H = -395.2kJ (note that we don't say "per mole" which would be ambigous, rather just leave it as is and know that it means for the equation as written)Īdding: S + O2 + SO2. Now you can write your equations and add them: Why: the sign flips when you flip the equation, and the enthalpy number drops by 0.5 when you scale the reaction by 0.5. Turn around your second input equation, and multiply it throughout by the factor (1/2): 
But you have that SO3 on the right! So, prepare to get rid of it by making it appear on the left in an equation, so as to cancel the one on the right when you add the 2 equations. Now, are you lacking any reactants on the left? Not really. So pick an equation from your input list with an S on the left: Look at your desired equation: S + O2 = SO2 There are very many routes to accomplish the same thing here's one of them. You want to add your equations so that you get your intended final equation, right?
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
